Share On Social!
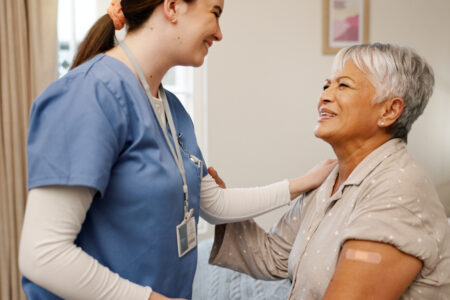
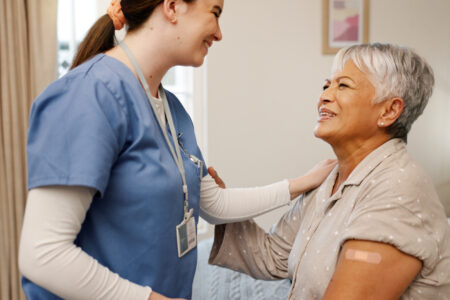
Recently, experts from the National Institutes of Health joined our Salud Talks podcast to discuss the COVID-19 vaccine and the “scientific pathway” that ensures safety for those who receive such treatment.
Now we’re talking all about the design of vaccine clinical trials and how they’re conducted.
Listen to the Salud Talks Podcast, Episode 36: “Understanding COVID-19, Part 2; Distribution of a Safe Vaccine,” as we dive into how researchers bring scientific discoveries, like COVID-19 treatments and vaccines, to their patients in everyday practice!
- WHAT: A #SaludTalks discussion about the COVID-19 vaccine and its distribution. We also discuss why people choose to take part in clinical trials and partner with researchers to share their unique, lived experiences to help advance science.
- WHERE: Available wherever fine podcasts are downloaded, including Apple Podcasts, Spotify, SoundCloud, Tune In, and others
- WHEN: The episode went live on July 14, 2021

- WHO: Salud America! Podcast Producer Josh McCormack; Dr. Katya Corado, a researcher with The Lundquist Institute; Dr. Jaime G. Deville, a safety review board member with University of California Los Angeles; Dr. Felix M. Valbuena, a medical care provider with Community Health and Social Service Center; and Emelina Asto-Flores, a COVID-19 Vaccine clinical trial participant.
In this episode, we explore questions such as:
- How are medical experts addressing patients’ concerns about getting the vaccine?
- What made a COVID-19 vaccine clinical trial participant decide to join?
- How can listeners have conversations with family and friends about the vaccine?
Be sure to use the hashtag #SaludTalks to join the conversation on Twitter, Facebook, and Instagram — share more information, stories, and advice about thinking and learning!
Don’t forget to check out the Salud Talks podcast, and, of course, subscribe on all platforms!
Click here to learn about Salud America!’s “Salud Talks” podcast, see upcoming and past episodes, and see how you can get involved.
Explore More:
VaccinesBy The Numbers
142
Percent
Expected rise in Latino cancer cases in coming years