Share On Social!
Liver cancer, one of the fastest growing causes of cancer-related deaths in the US, affects many people in the South Texas area with its large Latino population.
That is why researchers are working hard to improve early detection of liver cancer.
Exact Sciences, the company responsible for Cologuard, a popular brand of colorectal at-home testing kits, announced new findings on its new blood test that aims to detect early hepatocellular carcinoma (HCC), which is a common form of liver cancer, according to a company news release.
Let’s explore the new findings can benefit the health of Latinos and all people.
Early Liver Cancer Detection
Exact Sciences’ liver cancer blood test, Oncoguard, has been in clinical testing.
The new test results show that the product has proven to be a reliable early detection tool that has the potential to outpace current standard of care ultrasound imaging.
Dr. Binu John, principal investigator for the ALTUS trial, associate professor at the University of Miami Miller School of Medicine, said that ultrasound surveillance has been the standard for liver cancer screening for decades. But it’s limited by image quality and inconsistent follow up, resulting in low detection rates and poor adherence.
“A highly sensitive blood-based alternative like Oncoguard Liver is a game changer that could make liver cancer screening more accessible, [fair], and effective for millions of at-risk patients,” John said in the news release.
The laboratory conducted a study of the blood test with more than 3,000 participants of multiple backgrounds and populations, according to the news release.
What they found was that the new blood test could detect three times more cancers in their early stages compared to ultrasound imagining (67% vs 22%).
With findings like that, the company has shown that this latest technology could catch liver cancer early, when it’s at its most treatable.
“These new data from the ALTUS study show that we can reliably detect liver cancer earlier, which is key to improving outcomes,” said Dr. Paul Limburg, chief medical officer, screening, at Exact Sciences.
Liver Cancer in Latinos and All People
Liver cancer is expected to become the third-leading cause of cancer death, with more than 40,000 new cases and 30,000 deaths each year.
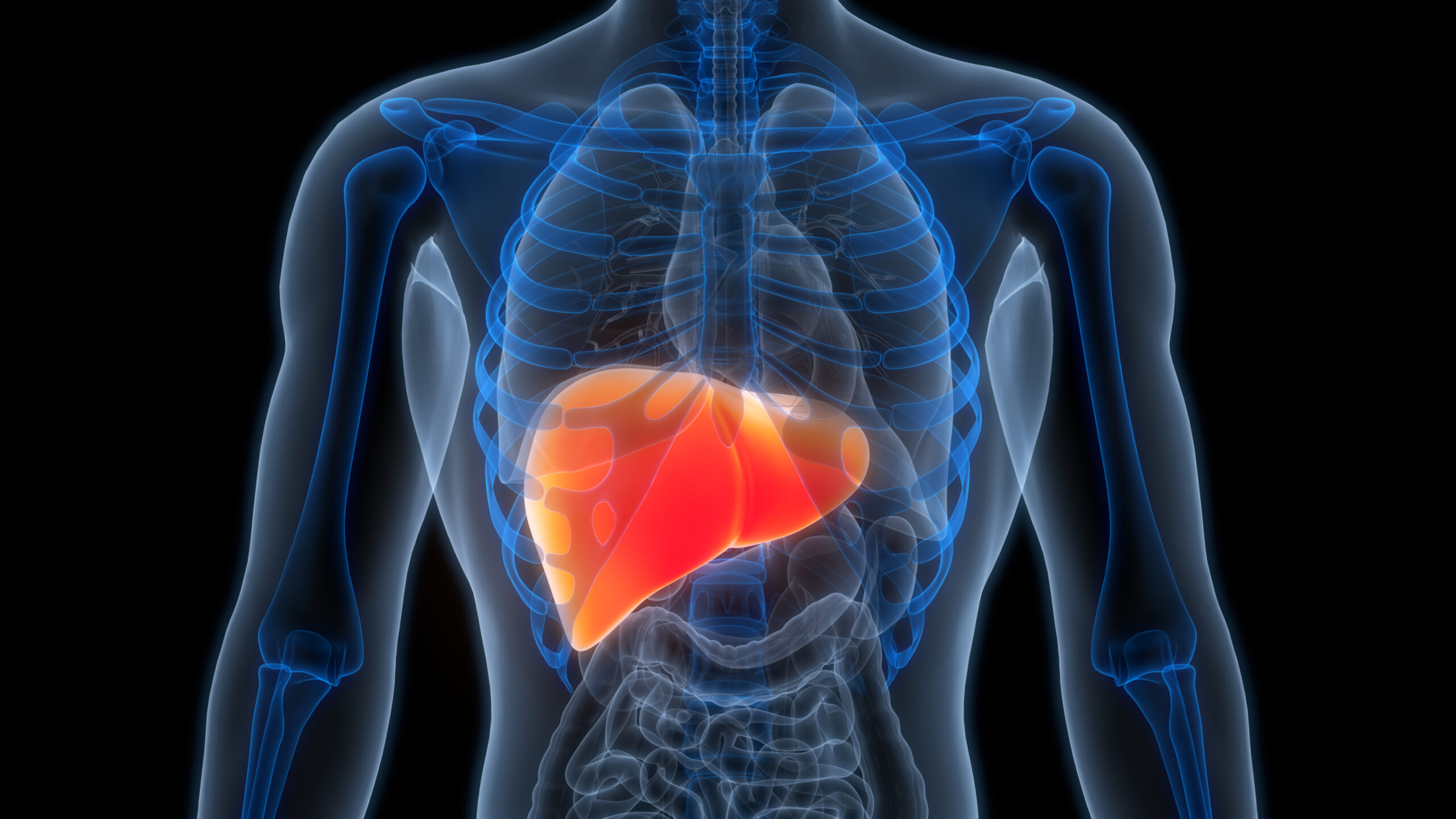
Those who are diagnosed at an earlier stage have a greater chance of survival when compared to regular surveillance – 12 times higher to be exact.
However, there is a distinct lack of participation in regular surveillance, with only 30% seeking it as a form of detection and prevention.
Regular surveillance is recommended for people who suffer from chronic liver disease and cirrhosis, which can be caused by alcoholic and non-alcoholic fatty liver disease.
Many included in this at-risk population are Latino due to a higher prevalence of obesity and diabetes.
Overall, 1 in 5 people with liver cancer in the United States are Latino, with an 4.7% increase since 2000, according to 2018 data.
Latinos are also more likely to be diagnosed at a later stage, making liver cancer deaths more prominent among the Latino population.
According to OMH, Latino men and women are 1.5 and 1.6 times more likely to die from liver cancer than their white counterparts.
That’s because vulnerable populations such as Latinos are more likely to lack cancer prevention and early detection resources such as access to oncology centers, transportation, and insurance.
With the new blood test, Exact Sciences is hoping to help close the gap on health inconsistencies in Latinos and all people and create more cancer survivors.
About Exact Sciences
Exact Sciences is a laboratory paving the way for new cancer screening and diagnostic testing, especially for cancer.
“Exact Sciences helps patients and health care providers make timely, informed decisions before, during, and after a cancer diagnosis,” according to the news release.
They do this through products such as Cologuard and Oncotype DX, Cancerguard, which tests for multiple types of early cancer detection, and Oncodetect, which tests for molecular residual disease and recurrence monitoring.
Exact Sciences was also a primary sponsor for the first ever Latino Cancer Patient Advocate Training Program, which convened ahead of the 2024 Advancing Cancer Research for Latinos and All Populations Conference.
The next conference is set for Feb. 18-20, 2026, in San Antonio.
Volunteer for a Clinical Trial
None of these innovations are possible without the contribution of cancer patients and survivors who helped the company test these products through clinical trials.
Clinical trials are studies that help researchers learn more to help slow, manage, and treat cancer and Alzheimer’s for current and future family members. But without volunteers for clinical trials, the benefits may miss this group.
When it comes to clinical trials, it’s important to have a participation population that aligns closely with the population of the US. That means having a spectrum of people from all types of backgrounds and walks of life.
This helps researchers make sure that these products, treatments, preventions, and interventions work for everyone.
“Volunteers in clinical trials are not only helping themselves, but they’re also building a future with better treatments that can help their families in the future,” said Dr. Amelie Ramirez, director of Salud America! and the Institute for Health Promotion Research at UT Health San Antonio.
Visit our clinical trials page to find a clinical trial, read about hero volunteers, and more!
Those looking for opportunities based in San Antonio can search the Mays Cancer Center at UT Health San Antonio’s Find a Clinical Trial database to learn more about available clinical trials and eligibility requirements.
On a national level, visit clinicaltrials.gov to find a clinical trial near you.
LEARN MORE ABOUT CLINICAL TRIALS!
Share Your Liver Cancer Story
Liver cancer survivors living in South Texas can share their stories with the Avazando Caminos Hispanic/Latino Cancer Survivorship Cohort Study.
The research study at UT Health San Antonio is looking to enroll 1,500 Hispanic/Latino cancer survivors in South Texas.
Participants must have completed primary cancer treatment for breast, lung, liver, stomach, colorectal, kidney, prostate, and uterine cervical cancers in the last 10 years.
Recently, the study announced it would now be enrolling participants who have completed treatment for bladder, melanoma, ovarian, thyroid, and lymphoma cancers.
The study is comprised of seven study visits over five years, which consists of assessment interviews, and some blood draws.
Volunteers are eligible to receive $50 per visit.
To inquire about volunteering for Avanzando Caminos, please contact a member of the study team at the Institute for Health Promotion Research at UT Health San Antonio at 210-562-6514 or email caminos@uthscsa.edu.
You can also visit the Avanzando Caminos website in English or Spanish for more information or fill out a short eligibility survey and a member of the study team will be in touch.
Join the Avanzando Caminos study in English or Spanish.
Explore More:
Latino CancerBy The Numbers
25
years
of life expectancy between some U.S. cities.



